Overview
The myTXTL Toolbox 2.0 Plasmid Collection was designed and intensively studied by Vincent Noireaux, PhD at the University of Minnesota, and co-workers. It contains more than 100 plasmids with various promoters and open reading frames (ORFs) to investigate gene regulation and molecule turnover. Available ORFs include a wide selection of transcription factors, TXTL modulators, and fluorescent reporter proteins to build multi-stage gene circuits.
A gene circuit executed in any myTXTL kit is required to start with a σ70-specific promoter, as gene expression in myTXTL relies entirely on the endogenous TXTL machinery of E. coli. Its modular and pre-designed format makes it ideal for training students and demonstrating basic concepts of synthetic biology.
NOTE: Toolbox 2.0 plasmids are intended for propagation purposes only. To make a plasmid directly suitable for in vitro expression in myTXTL, follow the recommended two-step procedure to generate high-purity plasmid samples. For propagation of P70a-vector, please read the Tech Note, Preparation of chemo-competent KL740 cells for amplification of P70a vectors.
Applications
The combination of synthetic gene networks with in vitro protein production technology opens new innovative fields of application in medicine and biotechnology, including:
- Environmental sensors
- Controls for biomanufacturing of biofuels
- Stem cell medicine
- Gene therapy (CRISPR)
- Manufacturing of functional materials
Cell-free platforms help to overcome limitations occurring in living host organisms, which are typically tied to the biochemical cross-talk between the host cell and the artificial gene circuit
and a consequence of the potentially cytotoxic effect of the exogenous gene network on the living cell on the one hand, and the availability of resources for transcription and translation, DNA replication and metabolites on the other. As those resources are dependent on the cell density, growth rate and cultivation conditions, deploying cell-free systems for gene circuit testing creates a more controlled environment, increasing reproducibility and robustness of the circuit behavior and output.
Performance
The myTXTL Toolbox 2.0 Plasmid Collection serves as a comprehensive source to build a variety of gene circuits executable in the myTXTL system. The examples below highlight selected features and capabilities. For more inspiration, visit our publication page.
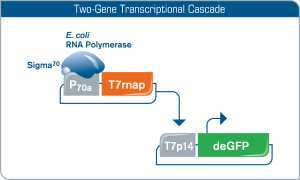
Utilizing a two-gene transcriptional cascade for T7 promoter expression
To facilitate T7 promoter regulated gene expression in myTXTL, T7 RNA polymerase is co-expressed from a helper plasmid (P70a-T7rnap) encoding a Sigma70-specific promoter compatible with myTXTL Master Mixes. T7 RNA polymerase then facilitates gene expression on the T7 promoter DNA template (here: T7p14-deGFP plasmid). Constitutive and inducible T7 promoter versions are acceptable.
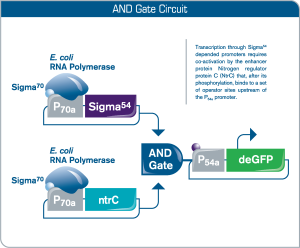
Execution of biologic logic gates in myTXTL
In synthetic biology, logic gates research is aimed at the development of decision-making gene networks for environmental and medical applications, in which biological gates act as sensors, detection units and drug vehicles. In this AND gate circuit, deGFP expression requires both phosphorylation of Nitrogen regulator protein C (NtrC) as well as presence of transcription factor Sigma54.
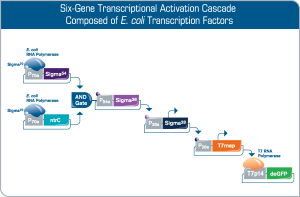
Rapid prototyping of synthetic gene networks
Engineering of synthetic gene networks involves executing an iterative design-build-test cycle which is accelerated by cell-free expression technology like myTXTL. By utilizing the native E. coli TXTL machinery, myTXTL gives access to an extraordinarily large repertoire of genetic building blocks. Here, a multi-stage cascade composed of a logic AND gate and bacterial transcription factors is producing a fluorescent output. Other fluorescent proteins are part of the myTXTL Toolbox 2.0 Plasmid Collection.
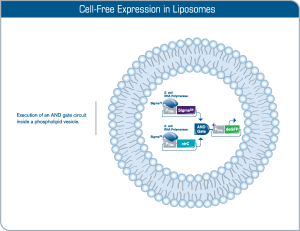
Compartmentalization of cell-free reaction
The encapsulation of myTXTL reactions into large phospholipid vesicles (liposomes) is an example of how artificial cells hosting cellular functions and complexity as advanced as in nature are constructed. Incorporation of transmembrane proteins provides more sensing options and facilitates cellular communication. Other approaches of compartmentalization such as emulsions, liquid-liquid phase separation, hydrogels and polymer-containing membrane compartments can also be applied.
Resources
FAQs
Transformation efficiency depends on the quality of the competent cells. Make sure that cells were immediately frozen after preparation and stored at ≤ 80 °C. Please also note that for some cells, transformation efficiency drops drastically over time. Additionally, we advise to use E. coli strain KL740 for amplification of any plasmids containing σ70-specific promoter like P70a.
No. All our Toolbox 2.0 plasmids (except the positive control plasmid P70a-deGFP that comes with the myTXTL kit are meant for plasmid amplification in E. coli only. The degree of purity is NOT sufficient for efficient in vitro production. Please refer to the current myTXTL handbook for recommendations on preparation of plasmid templates for myTXTL reactions.
All P70 promoters originate from the lambda phage promoter for the repressor Cro with its two operator sites and are specific to the E. coli sigma factor 70. They differ in strength (P70a > P70d > P70b > P70c) due to mutations that were introduced at -35 and/or -10 regions.
Yes, please see examples in the Publications section; filter for myTXTL. Please note, that every gene circuit should start with a σ70-specific promoter like P70a.
Efficient in vitro protein production is highly dependent on the quality of the template DNA, which should be free of nucleases (DNases, RNases) and inhibitors of the TXTL machinery (e.g. EDTA, ethidium bromide, SDS, Cl- ions, ethanol). Preparation of plasmid DNA with standard commercial kits usually involves sample treatment with RNase, which may not be completely removed during downstream processing. Thus, we strongly recommend subjecting the prepared DNA to either a commercial PCR clean-up kit or standard phenol-chloroform extraction and ethanol precipitation. Ideally, template DNA is suspended in nuclease-free water. Please note, introducing Mg2+and K+ ions can compromise the kit performance, as they are extremely critical for transcription and translation, and are optimized in the master mix.
Yes. For all plasmids containing the lambda phage promoter (P70a, P70b, P70c, P70d) it is extremely crucial to use E. coli KL740 as the transformation strain. When cultivated below 30°C, this strain over-expresses the lambda phage repressor protein Cl857 that represses P70 promoters, thus ensuring high transformation efficiency and plasmid stability. KL740 can be purchased from E. coli Genetic Stock Center (Yale) [CGSC#: 4382] or from Daicel Arbor. For all other plasmids, a standard laboratory E. coli cloning strain like JM109 or DH5alpha is sufficient.